|
light (read wavelengths) in the dispersing beam. Flame microsampling is accomplished using a micropipet to place 50–250 μL of sample in a Teflon funnel connected to the nebulizer, or by dipping the nebulizer tubing into the sample for a short time. It can also be used to isolate a single line from a light source such as a hollow cathode lamp. The source for atomic absorption is a hollow cathode lamp. With an Agilent multi element hollow cathode lamp, one lamp can be used for determination of multiple elements, avoiding lamp changes during analysis and extending the number of elements you can determine in the same analysis. Note that the peaks in the top row have the same width thus, the x-axis scale is different for the continuum spectra than for the line source spectra. For example, continuously aspirating a sample that has a high concentration of dissolved solids-sea water, for example, comes to mind-may build-up a solid de- posit on the burner head that obstructs the flame and that lowers the absorbance. 1: Illustration showing the effect on absorbance of using a continuum source and a line source for atomic absorption. Aluminum (Al) hollow cathode lamp has a Primary Wavelength of 396.2 nm (secondary wavelengths 308.2-309.3). Continuous aspiration is sample intensive, typically requiring from 2–5 mL of sample.įlame microsampling allows us to introduce a discrete sample of fixed volume, and is useful if we have a limited amount of sample or when the sample’s matrix is incompatible with the flame atomizer. The most common means for introducing a sample into a flame atomizer is a continuous aspiration in which the sample flows through the burner while we monitor absorbance. 
Hollow Cathode Lamp Wavelength Rs 15,000 Usage/. The relative size of each zone depends on many factors, including the choice of fuel and oxidant, and their relative proportions. Hollow Cathode Lamp Rs 25,000 Material: Aluminium Hollow Cathode Lamp Wavelength. From: Vacuum Deposition onto Webs, Films and Foils (Second Edition), 2011. The light produced by the lamp is emitted from excited atoms of the same element that is to be determined, therefore the radiation energy corresponds directly to the. Profile of typical flame using a slot burner. In atomic absorption spectroscopy, the light from a hollow cathode lamp or laser is partially absorbed by the gas-phase atoms and the remaining light is passed through a monochromator and into a detector. The free atoms are then exposed to light, typically produced by a hollow-cathode lamp, and undergo electronic transitions from the ground state to excited electronic states. A low pressure of argon in the lamp insures that the line width from the hollow cathode lamp is less than the line width of the absorbing species.\). Collisions of excited state sputtered atoms with argon atoms will lead to broadening of the output of the hollow cathode lamp and potentially lead to the same problem described above with the use of a continuum source. The pressure of the argon is low to minimize collisions of argon atoms with sputtered atoms. With this understanding we can ask why the hollow cathode lamp has a low pressure of argon filler gas. 
Therefore, hollow cathode lamps, which emit intense narrow lines of radiation specific to the element being analyzed, are needed for atomic absorption measurements. Reducing the slit width on a continuum source to a level that would provide a narrow enough line to respond to atomic absorption would reduce the power so that it would not be much above the noise. Applying a potential across the electrodes ionizes the filler gas. The problem with reducing the slit width is that it reduces the number of photons or source power reaching the sample. The source for atomic absorption is a hollow cathode lamp that consists of a cathode and anode enclosed within a glass tube filled with a low pressure of an inert gas, such as Ne or Ar (Figure 10.4.5 10.4. What is the problem with reducing the slit width of the monochromator to get a narrower line? 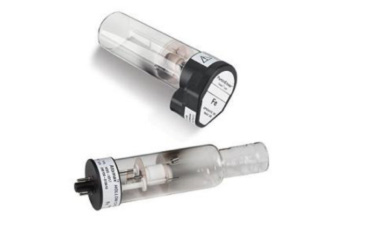
Atomic absorption line superimposed onto the overall output of a continuum source/monochromator system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
